Simplyfing Complex Science for FDA Success
The Challenge:
CraniUS Therapeutics needed an animation and technical schematics to clearly communicate their device’s mechanism and intended use for a pre-submission meeting with the Food and Drug Administration (FDA). Their challenge was simplifying a highly complex product for regulators while ensuring that the principles of operation, fit, and function were conveyed effectively.
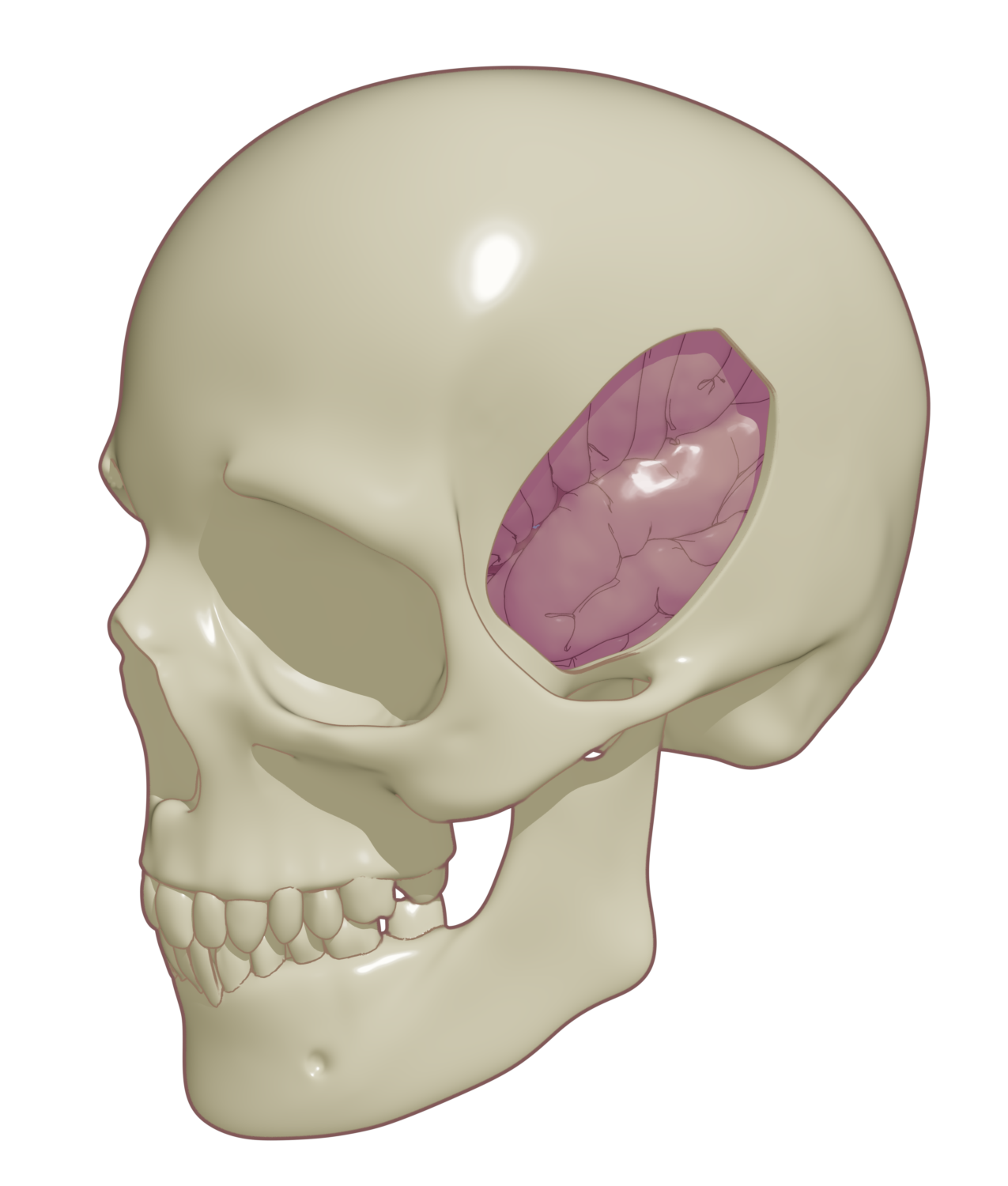
The Impact:
With these assets, CraniUS effectively communicated the device’s functionality and intended outcomes during their FDA meeting. This resulted in a productive discussion and a clear understanding of the most likely regulatory pathway for their device.
Capabilities Delivered:
- Concept development and animation
- Technical schematic design
- Principle of operation documentation
- Regulatory communication support

